At this point the mushroom cloud of the Tzar Bomba had reached an altitude of 56 kilometers. The narrow area of the base is where the 10 kilometers of fireball detonated.
For those who have missed the previous 3 parts and the prequel. Here are the links.
Laki deconstructed: I. prequel
Laki deconstructed II. Anatomy of an Eruption
Laki deconstructed III. Grimsvötn and Beyond
Laki deconstructed IV. A Timeline for Destruction
The nuclear background
To be able to understand large eruptions you need to understand a bit about nuclear weapons. In this case there are several reasons why we need to do analogies with the darker side of physics and the volcanoes that we love to learn about.
But first we have to become archaeologists. Some of you might have noticed that when using carbon-14 dating you get a result with the time set to BP (Before Present), and if you have studied archaeology you will know that this means the year 1950. One might think this is just an arbitrary date, but it is not. This is the last year where you can do any atomic dating due to nuclear fallout from aerial nuclear weapons testing. It will take about 100 more years before it is possible to test our present time. Imagine, from the year 1950 up to 2100 there will forever be a hole in the chronology.
Now, those with a bit of pangeant for Cold War history will believe that the spike in the picture above comes from the Tzar Bomba test, nothing could be more false. The Russian Sakharov-designed nuclear bombs were much better designs than the American Ulam-Teller bombs. The Tzar Bomba used up 98 percent of the available fissionable material, the largest American bomb (Castle Bravo) only used up 13 percent. For all points and purposes every American hydrogen bomb up until the late sixties was a dud, albeit effective enough to kill people on a horrifying scale.
The highest point of nuclear material fallout is in 1963, and that date is one of the two used to calibrate ice core samples. It is easy enough to find. The other year used to calibrate the measurements is the Lakí event. The main reason for this is that those two show up in every ice core drill-sample in the northern hemisphere. It is a pretty handy tool for scientific comparisons between different glaciers and for calibrating timelines within the respective ice core.
The Lakí fallout trace is in two parts. First you will find an ash layer, the second part will be emplaced on top of the ash, and that is a sulphate layer, residual acidity from the sulphurous gasses released by the eruption that both stayed airborne for a longer period, and also was released up to a year after the eruption had stopped.

One of the main glaciers at Svalbard in Spitsbergen
Calculating Nuclear Fallout
As any good scientists Volcanologists steal what they need from other scientific disciplines. In physics we steal a lot from the mathematical department. So, it is befitting that the Volcanologists raided us for the formulations to calculate nuclear fallout patterns when they needed to calculate volcanic fallout. The difference is normally so small that it should not matter really. But there are differences, some small and some so large that it becomes a problem.
First of all, as a physicist we differentiate nuclear dust (any dust that became radioactive during the detonation) and true radioactive byproducts from the detonation. The first part will behave like your average volcanic ash. It will drift around for a while depending on size and how high it was lofted into the atmosphere. The second part is basically atoms of fusionable or fissionable material that was not used up in the detonation. This will drift for a much longer time.
This is not so important really; it will though give a negative margin of error. Volcanic fallout normally travels shorter than the model gives at hand since you do not have traceable individual atoms.
There is though a real problem when using the model. The basic model was developed for the original small scale fission bombs used over Hiroshima and Nagasaki. When we started to detonate the far larger hydrogen-bombs it was rapidly discovered that the formulations had less and less with reality to do. This was foremost a problem with the dirty American Ulam-Teller bombs.
Let us now for a moment get back to the picture in the beginning. The top of the mushroom cloud is at 56 kilometers height, and that is a full stratospheric injection of fallout material. At those heights any particles will pass several times around the globe before they drift to the surface.
We now have to answer a question. What is it that lifts ash up into the higher parts of the atmosphere? Most people believe it is the explosive force that lifts ashes up into the atmosphere, but that is not true. What lift the ashes are convective currents of air. In simpler terms, ashes ride upwards on the hot air rising.

The fireball of the Tzar Bomba is actually a 10 kilometer wide sun. “I have become Death, destroyer of worlds”.
The Tzar Bomba was an almost instantaneous event where a fireball 10 km across was created, and it was at its hottest as hot as the core of the star. In an instant flash of terrible beauty humanity had made a star on planet Earth. It created a cloud of ash that rose higher than anything in human history, but it contained only what it lifted during the first few minutes, and for a couple of months it drifted across the entire northern hemisphere before falling down, almost uniformly spread.
A volcano has a much lower power output than that, but if the volcano is big enough, and heats a large enough area and the eruption last for a sufficient time it will sooner or later rival, or even surpass, the amount of ash ejected into the higher atmosphere. The height is more limited, however.
From Ashes to Ashes
The Skaftár Fires in its initial stage caused an 1800 meter high fire curtain, and it also heated a large area. This 1200 degree air and gasses sucked in air from all sides and created a tremendous upwelling of heated gas and air mixture. This created a bubble of denser than normal air to form above the eruption and inside the bubble, ash, dust, and shards of tephra was lifted. Anything heavy quickly fell down again as rubble, but the finer particles smaller than 2 millimeter hung there suspended at an altitude ranging from 13 to 18 kilometers, reaching the upper troposphere and lower stratosphere. The suspended particles were then moved onwards on the higher air currents at high speed.
The particles then quickly started to fall downwards as the heated air and gas cooled at altitude, and they fell according to size and weight. What is interesting is the time it took. Before this it was considered to have happened rather fast and according to the basic function of the nuclear fallout model due to the assumption that the atmospheric injection stopped at a maximum altitude of 10 km (boundary level between troposphere and stratosphere).

From Ashes to Ashes and from Dust till Dusk. Note the face mask and social distancing.
How do we then know that the ashes reached a higher altitude? We know this from the fact that we can find ashes in ice core samples across the northern hemisphere. The difference is that the heavier and larger particles are gone in the Greenland samples, but not entirely in the Svalbard samples. Incidentally, that also gives us the direction it travelled.
We know that the particle count in Svalbard is on average 250 particles per square meter, and that sums up to less than a gram per square meter. That does not sound a lot, until you start a bit of calculation. Now you say that less than 1 gram per square meter is not a lot to hang up in a Christmas tree. Only problem here… there are a lot of square meters if you sum up the polar zone and the temperate zone in the northern hemisphere. This is not a mathematics site, so the heavy math was done for you on Mathematica. The result is that the weight given by the mathematical model is 255 000 000 000 kg. With an average density of 3 000 that gives 0.085 cubic kilometers of rock injected high in the troposphere and lower stratosphere, recalculated into dense rock equivalent. Please note, this estimate assumes a uniform distribution of ash. In practice, close to eruption there would have been much more, and further away much less. But some of the ash stayed aloft long enough to circle the globe and end up in the Greenland ice. Fragments have been found in Ireland. During the eruption, ash falls were reported from the Faeroe Islands.
It is fairly uncommon to find volcanic tephra in ice cores in Greenland. The ash that is present is mostly from rhyolitic, explosive eruptions. The Öraefajökull AD 1362 eruption covered Greenland in ash. In contrast, ice-core tephra from basaltic eruptions is extremely rare: Laki was exceptional in managing to deposit some, and even for Laki, only a few fragments have been found (But this dominance of rhyolitic ash is only true for the holocene. Tephra in Greenland from the ice age is mostly from basaltic eruptions! Whether this was due to different atmospheric circulation or different eruption styles in Iceland is not well known.)
From sulphates to sulphates

Killed by acid rain
The ash did not stay aloft for long. But now came the next pollutant. After the ash came the smell. In the Svalbard ice core mentioned above, the ash is the lowest level, and above it is a layer of sulphate particles. These came down as late as 6 to 12 months after the ash. This delay is for two reasons. Most of the high-altitude ash was ejected early on in the eruption when the eruption rate was highest. The later events also started explosively, but they were not as energetic. Therefore, the ash was lifted less high and came down closer to the vent. One reason for the initial explosions, by the way, is that the graben where Laki erupted was wet. There may even have been lakes. The lava erupted into water, with predictable results. Later in each eruption the water had evaporated, explosions ceased and effusion took over.
But sulphur is continuously emitted while lava flows, changing to sulphate in the rising plume. The sulphate ebbed and flowed with the rest of the eruption, but it kept going. Some of the sulphate stayed in the lower troposphere and mainly affected Iceland. But some went much higher, in the upper troposphere and lower stratosphere. These aerosols stayed aloft far more easily than the ash, and the sulphate kept coming long after the ash had gone. And so, the sulphate traveled easier and lasted longer. Some even reached the southern hemisphere. There was still significant sulphate over 10 kilometers up during early 1784. As Laki waned, so did the sulphate: very little was emitted after November 1783. But for 5 months, the North Atlantic was engulfed in a toxic haze. High latitude eruptions may be worse than tropical ones: they spread their gas over a smaller area (the northern temperate zone is smaller than the tropics) and so can give high concentrations. But Laki would have been bad either way: this was not a normal eruption: it was a mini flood basalt. The gas output was enormous.
Iceland itself was very badly affected. The sulphate fog killed the grass, and fluorine killed the cattle. Acid rain burned holes in leaves and left skin wounds on people and animals. Birch trees died across Iceland. 80 percent of sheep and half of all cattle died. This caused a lack of food: it is called Iceland’s haze famine. And it was followed by one of the worst winters Iceland has ever had. The population of Iceland declined by 21% because of this.
The haze was quickly noticed in Western Europe: ‘On many days after the 24th June, in both the town of Groningen and countryside there was a strong, persistent fog … very dense and accompanied by a very strong smell of sulphur.., many people in the open air experienced an uncomfortable pressure, headaches and experienced difficulty breathing‘. (Brugman) The leaves fell off the trees, making it look like October rather than June. Parish records show a significant excess death rate that summer in the east of England: more people may have died from Laki in England than in Iceland. However, the timing suggests this was not due so much to the bad air but rather to diseases later that summer and during the exceptionally cold winter. Europe’s death toll is an indirect rather than direct effect from Laki.

From Thordarson and Self 2001
The haze appeared in mid June. There was high pressure over central Europe. The sulphate had been transported in the upper troposphere by the wind, and the downward airflow in the high pressure brought it to ground level. By July, the haze covered much of the temperate northern hemisphere, including China and Alaska. A second wave of the ‘dry fog’ came in August.
The effect on the climate was complex. Europe had heat and drought, with temperatures 3 degrees above normal. Unusually severe lightning was reported from England in July and August. But Iceland was cold, Moscow had June snow and Japan had such cold weather that the rice harvest was affected. Sulphate haze absorbs sunlight. If the sulphate is at ground level, this can heat the air and cause higher temperatures. This may be what happened in Europe, although it is clear that the atmospheric circulation was also important. The blocking high over Europe may even have been unrelated to Laki. But stratospheric sulphate heats the stratosphere, and as light can only heat once, the ground cools. And indeed, even in Europe and Eastern North America, after the heat came the cold. The winter of 1784 was extreme. As Laki faded, Europe froze.
Carl and Albert




this has been a fascinating series, made me think of some of the rocks I have been collecting< I do live in a volcanic field, active a long time ago
Lovely and do we have the actual temperature readings from that winter of 1784? Best!motsfo
There’s the long term Central England Temperature records at https://www.metoffice.gov.uk/hadobs/hadcet/
You can also download daily, monthly and seasonal averages at tne above page.
Nice article. I just wonder. These rifting events happen approx. every 270-300 years and are devestating to Iceland. Is it possible to create a rifting event with a conventional bomb? If so, would it give less damage than the next rifting event in 30 to 70 years? Wonderful theme for ethics and geoengineering people and of course the population living ‘nearby’.
No, they cannot be generated from above. It is rifted by the stress and pressure from below. You can get rifting without an eruption, but that is elsewhere in Iceland. In this region, the rifting event is invariably triggered by magma.
Lohi picking up
50 quakes all magnitude in Loihi region last 24 hours
Largest
3.8 48km SE of Pahala, Hawaii 2020-05-12 03:58:09 (UTC) 6.9 km
Question from one of Volcano Cafe’s more dumb followers: is the Loihi Seamount likely to be the “next” upcoming island in the chain, or an extension to Hawaii? Or is that unlikely? Thanks!
It is already a decent mount. Yes, it is likely the next volcano. I would guess it will become connected to the Big Island. As volcanoes grow and sink, exactly which ones define which island is changeable. Perhaps Hawaii always grows like this: connected volcanoes, disintegrating into separate islands as they sink.
Usually it grows by connected volcanoes, particularly when the hotspot is strong, But there can be jumps, for example Haleakala never joined subaerially with the next volcanoes in line, Mahukona and Kohala, so that Maui and the Big Island never formed part of the same land. But that is the only time it has happened in the last 5-4 million years (since Kaena volcano).
As I understand it, ‘sorta both’. Loihi looks too close to become a stand-alone island. By the time it surfaces, it will merge with the existing island. Of course, if eruption rate is too slow, the ‘hot-spot’ may have moved on, leaving Loihi as a shallow sea-mount to NW of possible Loihi Jr..
Meanwhile, erosion and flank mega-failures and such nibble away at the existing island chain, whittling them down. Having lost their hot-rock uplifts, entire island subsides into their isostatic moat, now partly filled by debris from those flank collapses…
( I don’t understand where/how/why ‘rejuvenation’ eruptions pop up, but they may considerably extend their islands’ life-time…)
So, if Loiho makes it to the surface and, merging with main island, builds a nice shield, it will probably be there when rest of island has eroded to remnants and, off to SE, Loihi Jr is developing…
Thanks!
I have to wonder – does HVO maintain a hydrophone network on Loihi to listen for signs of eruptions?
They have at least once placed some equipment on Loihi during previous events but funds didn’t permit permanent operation.
USGS has tweeted this
https://twitter.com/USGSVolcanoes/status/1260033262916734977
There’s also a status update at https://volcanoes.usgs.gov/volcanoes/loihi/status.html
The laki lavas where erupted at very fast eruptive rates.. and flowed quickly on open channels.
The drained lava channels there, suggest it was a very fluid eruption indeed.
Whats the estimated viscosity of Laki lavas in Pa.S?
I know there is a near-zero chance that people misunderstood Jesper’s question, but he could of been referring to the volcano, Laki, not the fissure, Lakagigar. Is it worse to call the event a “fire” or name it after an older volcano that the fissure passed through?
A ‘fire’ is actually the adopted name for an Icelandic style rift eruption. It is no worse than calling the whole thing a ‘vulcano’ which is actually the name of an Italian island.. But yes, ‘Laki’ is a bit of an unfortunate name for the event.
And obviously no one measured Laki’s viscosity during the eruption, and there is not much point measuring it for solidified rock. But assuming 1200 C, it may have been around 50 Pa s.
Laki flowed like raging rivers like spring river in full flood…
Huge open lava rivers flowing very very quickly in open channels.
Skaftareldahraun lava flows flowed maybe
at 80 kilometers per hour? close to the vents?
Must have been a beautyful sight..
But tiny compared to Siberian Traps and Opening of the Atlantic
In well formed channels lavas can flow very quickly indeed.
Both Fissure 8 and Holuhraun flowed very quickly close to the vents
Laki was erupted on flat ground too
But I imagines a way higher lava flow speed with 6000 cubic meters a second.
Spectacular standing waves, currents, and turbulence Skaftareldahraun must have produced in its lava channels.
At 50 Pa.s it will be a very fluid eruption.
The lava channels in Laki are similar to Holuhraun and Leilani in textures.
How quickly flowed Skaftareldahraun in its channels?
Kilometers per hour I means
The flow expanded by 15 km per day.
It woud be massive Aa lava then .. moving at walking speed
Feed by very very fast moving fountain feed channels upslope
Right?
From HVO
https://volcanoes.usgs.gov/volcanoes/loihi/status.html
“INTERPRETATION AND HAZARD
This swarm may represent a brief magmatic intrusion or movement of magmatic fluids within the volcanic edifice . Although the swarm appears to have diminished in intensity, if earthquakes become shallower, it could lead to the beginning of a submarine eruption, similar to what occurred in 1996.
An eruption of Lōʻihi, if it were to occur, may cause partial draining of its summit magma chamber and summit collapse, as happened in 1996. Significant, sudden changes to the volcano’s surface could displace large volumes of ocean water, which, if large enough, might generate very small local tsunami waves . Earthquakes of magnitude 4 and above could occur if the swarm were to intensify and these may be felt on the Island of Hawaiʻi.
If an eruption or stronger earthquakes occur, very small tsunami waves may affect southeast shores of the Island of Hawaiʻi. Relatively low-energy, steam- and gas-driven explosions can occur at the depth of Lōʻihi, but with limited local effects on the volcano and surrounding ocean water.
There is no direct relationship between the current Lōʻihi swarm and the ongoing increased seismicity observed in Pāhala over the past year. The Lōʻihi swarm is ~35 km (21.7 mi) southeast of Pāhala, at significantly shallower depths. The current Lōʻihi swarm is also unrelated to seismicity observed on the south flank of Kīlauea.
“
Mila now has a Reykjanes webcam live: https://www.livefromiceland.is/webcams/reykjanes/
Sweet! Looks like it’s right on top of Mt Thorbjörn.
Thanks for the info. Now all they need to do is improve the Icelandic weather…!
And get someone to hack the cam to improve the turning speed… ha!
If you click on the left hand side bar, you can get 24 hours revolutions in a minute and a half, but I suspect you didn’t mean that fast!
Thanks for posting the link. Now we are ready for the big show, if it’ll take place.
Looks like the inflation has restarted at Mt Thorbjörn and neighbouring GPS stations:
https://notendur.hi.is/~hgeirs/iceland_gps/rnes/rnes_100p.html
Typo in article
No wonder the model didn’t work 🙂
Fission bombs over Hiroshima and Nagasaki
The ones you never heard about.. but you are right, and will be fixed.
Well bomb designer Ted Taylor did claim in his later years that there were ways of building simpler fusion devices he knew of. I don’t think he meant in 1945 though 🙂
He was the one behind the Davy Crockett warhead, Project Orion (mini atom bomb powered starship) and also famously once lit a cigarette with a 15 kiloton atomic bomb.
You may have already read it but if not I recommend
The Curve of Binding Energy Paperback – 1 April 1994
by John A. McPhee (Author)
http://awesci.com/nuclear-bomb-cigarette-lighter/
Sounds like a Robert Heinlein hero
The southwest rift zone of Mauna Loa is very noisy today.
And Campi Flegrei is continuing its build-up
I wonder if this almost 3 decade explosive eruption drought is the calm before an outbreak.
Tallis Rockwell ….. do you want Opening of the Atlantic 2.0 in Africa?
Is a insane – flood basalt your goal to flood to world with lava?
Looks like Tallis Rockwell wants to see whole continents buried by lava?
But I guess you wants Flegeri to go off like La Garita
But instead you may get something like Pinatubo 1991
Hi Folks,
My understanding is that the next Flegeri eruption is likely to be a Monte Nuovo scale event. Locally devastating but no regional impact as such. Has this thinking changed?
A previous (and awesome) article on this site played out a small scenario with regard to what this may be. Could a dragon or other do a fuller article on the, likely timing (volcanoes do what they like) 2020 was mentioned previously, size of eruption (pinatubo a realistic scale for next eruption?), run up to eruption, impacts on Naples, Italy or beyond. I note the endless supply of water in the area and extensive caldera.
Regards to all
Richie
Long time lerker, seldom poster
Hi Richie. Yes, Henrik’s article was great, and scary. It is worth noticing that (1) the chance of an eruption in a system that has seen only one event in several thousand years is not high, and this increase in activity may have happened many times before without any follow-on; (2) although this was in our list of super-erupters of the past (https://www.volcanocafe.org/power-of-the-past-25-super-eruptions/), nowadays the eruptions are minor. Still, a minor event inside a major city is not to be taken lightly. We have done a brief discussion in https://www.volcanocafe.org/up/ Perhaps it is time for a re-visit. I love Naples: it is a lively and down-to-earth. But not the area around the railway station where some of the main hotels are – that is another side of Naples.
Activity at Campi Flegrei is episodic, there have been three pulses (epochs) of eruptions since the caldera collapse:
https://www.researchgate.net/profile/Valerio_Acocella/publication/334812142/figure/fig1/AS:786826486898688@1564605461306/Eruptive-history-of-the-Campi-Flegrei-caldera-during-the-last-15-ka-A-Shaded-relief.ppm
Epoch 3 saw 8 VEI4s and a VEI5 eruption within ~850 years (according to VOGRIPA). Question is when will the next pulse happen and what controls them.
Jesper, Now I just want to see and track an eruption greater then VEI 4…If I can’t witness a VEI 8 or 7, then I would love to see and track a remote VEI 5 or 6. I refuse to raise any hope for a flood basalt because all of the biggest magma producers on the Earth probaby won’t be making an eruption of that scale any time soon.
Just looking at the seismology. Are there a couple of dykes in the area?
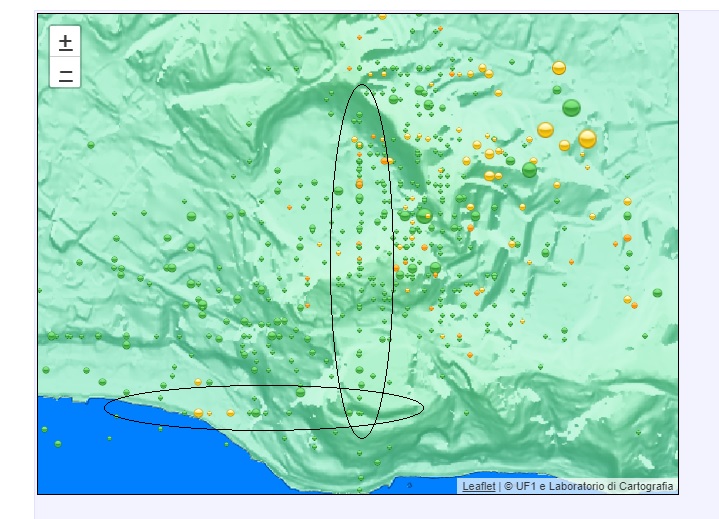
Campi Flegrei, that is…
I think those are quakes where the location could only be measured in one direction, not the other. The telling sign is that the line is very nicely vertical (or horizontal) on the chart.
Fair enough, thanks! I was mulling over the CF monitoring site and I see there has been a trend to a more magmatic origin of the gases since @2010. I suppose the volcano is just doing its regular ‘breathing’. Nonetheless, interesting to observe,
https://earthquake.usgs.gov/earthquakes/eventpage/nn00725272/executive
M 6.4 – 56km W of Tonopah, Nevada
2020-05-15 11:03:27 (UTC) 38.159°N 117.877°W 4.1 km depth
Looked it up on the USGS map… faults there don’t even have names.
I looked the area up on Smithsonian, those quakes are under what is known as the Aurora-Brodie volcanic field.
https://volcano.si.edu/volcano.cfm?vn=326802
Pleistocene era eruptions only but it is quite close to Mono Lake.
Trying again with Imgur
https://imgur.com/vbxVczQ
It would be interesting to have some eruptions in the Basin and Range. Lots of little monogenetic volcanic centers scattered around Utah and Nevada.
Don’t get my hopes up…Please!
Here is a screen shot with satellite imagery and today’s quakes
blob:https://imgur.com/e5241126-23dc-4b86-9132-faf999d560e8
not sure what happened there with Imgur.
Old Towns with brick buildings. i am familiar with that part of Nevada. The old west.Yet Tonopah has a mini Area 51 airbase there… Report are that the quake was at 4-5 km in depth..
Lots of strong aftershocks…
But it is that Nevada basin and range county- many faults…
It put a crack in the road.
Interesting University of Iceland research published today on precursors to eruptions and volcano plumbing, based on Bárðarbunga 2014-15.
Full article here but restricted access, I think: https://www.nature.com/articles/s41467-020-16054-6
Summary, with comments on BB, here: https://english.hi.is/news/new_understanding_of_magma_movements_in_volcano_roots
Hope everyone is keeping safe and well.
So Bardarbungas molten part of its magma chamber is a
10 kilometer wide and nearly
1 kilometers thick flat sill laccohith like body
Looks like a melt lens in a mostly mushy magma chamber region.
Bardarbungas 2014 magmas where very hot ( almost 1200 C ) and where very runny.
One of the hottest lavas ever seen in action.
So Bardarbunga should be pretty molten and open inside.
But is there any open conduit inside Bardarbungas depths?
And, again, what’s the relationship to Greip, which seems wide open? (Apologies: I know this has been discussed a lot on here but I haven’t been able to follow all the discussions, so doubtless I am out of date.)
nice little swarm near (outside rim) Törfajökull, cfr. IMO-website. Seems to be on the SW end of the Veidivötn Fissure Swarm?
The largest Hawaiian volcano – and it is not Mauna Loa
https://scitechdaily.com/puhahonu-revealed-largest-and-hottest-shield-volcano-on-earth/
https://en.wikipedia.org/wiki/Gardner_Pinnacles
Impressive!
Thats 200 000 km3
Really shows how powerful the Hawaiian hotspot really is
If the Hawaiian hotspot was under a very slow moving seafloor we woud end up with something larger than Tamu Massif
Katla Volcano is weaking up
https://www.volcanodiscovery.com/katla/news/103690/Katla-Volcano-Volcanic-Ash-Advisory-ERUPTION-TO-15KM-OBS-VA-DTG-151500Z-to-20000-ft-6100-m.html
Exercise only, I am afraid. Nothing happening. Now, if you looked at Greip, that does have a swarm.
https://www.google.se/amp/s/www.sciencealert.com/the-world-s-largest-volcano-isn-t-what-we-thought-it-was/amp
Albert look at this!
Looks like Hawaiis Pūhāhonu volcano had magma at 1700 degrees C
Archean hot magma.
Under the crust the hotspot was Around 1700 degrees C ( 3090F )
Looks like it had a major pulse in strenght back then.
The ongoing pulse may form similar magmas in the future knowing
How huge and long Kilauea already is
Repost
”Chemical analysis of rock collected from the volcano revealed a higher concentration of an olivine mineral called forsterite than we’ve ever seen in a Hawaiian volcano. This mineral indicates magma on the higher end of the temperature. calculations, the magma clocked in at 1,703 degrees Celsius (3,097 degrees Fahrenheit) – hotter than any other Hawaiian basalt ”
tsunami threat in Alaska
http://www.newsminer.com/alerts/scientists-potential-landslide-could-generate-large-tsunami-in-prince-william-sound/article_abf68376-963b-11ea-a36b-c37a684701b9.html
Double Indonesian action today. One FL450 puff from Ibu and an ongoing FL460 at Semuru.
https://watchers.news/2020/05/16/semeru-eruption-may-2020/
https://www.volcanodiscovery.com/volcanoes/today.html
I’m still not certain these are not false alarms. Can’t find any other details anywhere. No official word yet. 🤔
Semeru had an ash ejection but there was only a brief warning and it appears the ash quickly dissipated. It has been erupting: here is yesterday’s Sentinel image.
Lava flow?
The recent Tonopah M6.5 earthquake has triggered a very robust aftershock sequence, which now extends WSW-ENE over 30 miles (L) and 20 miles (W). Over the last 24hrs, there is also an apparent spike of scattered (induced??) earthquakes up to 150 miles away….including a spike in small earthquakes within the recent Ridgecrest M7.0 aftershock zone and near Mammoth Lakes (Long Valley). Not that that unusual for larger quakes (especially thrust events) to induce remote seismicity (sometimes thousands of miles away), but a M6.5 left-lateral strike slip doesn’t usually exhibit such behavior….unless there is an anomalously large area of direct stress transfer taking place? Curious.
This quake occurred in an area that has experienced many strong seismic shocks. A 6.1 quake struck in the area of the Columbus Salt Marsh in 1910. That location is near Coaldale and the junction of Hwy 6 and Rt 95.
More significant, was the somewhat famous 7.1 Cedar Mountain Earthquake in 1932. Cedar Mountain is north of the area of the Tonopah quake, and initiated a period of increased seismic activity that lasted a few years in the region. Wabuska, in 1933; and Excelsior Mountain, in 1934, were both in the 6.0 – 6.5 range, and occurred in the same general area, with the the strongest of them all being the 1915 quake at the north end of the Walker Lane seismic belt.
Check out this document for a general history of Nevada’s big quakes.
https://www.shakeout.org/nevada/downloads/NV_Earthquake_Hazard_History.pdf
The part of Nevada near Tonopah is in a very interesting seismic zone called Walker Lane, which is located along the western edge of the larger Basin and Range extension zone that basically extends from the eastern Sierra/Cascades to the western Rockies. The Walker Lane is a broad/broken region of faults trending SE-NW that roughly runs parallel to the Sierra Nevada, and is believed to be delineating the formation of a new continental boundary (transform fault) separating the Pacific and North American plates…in essence a new/developing San Andreas fault shifted 500 miles to the east. One of the things that’s unusual about the Walker Lane seismic zone is widespread presence of small (many unnamed) sub faults called Reidel shears that can generate earthquakes into the mid-6 magnitude.
It’s also worth mentioning, that there is some discussion going on in the backroom that in light of several potent swarms along with powerful main shocks that have hit the general vicinity of the upper Great Basin (near Boise, Salt Lake City, Wells, and several near Reno/Carson City in the last few years, that Nevada/Idaho/Utah may finally be returning to a more active period after being anomalously quiet for the last several decades.
Here’s an excerpt (and link) of a paper from 2008 that nicely goes into some of the history/background/future of the Walker Lane.
“In the western Great Basin of North America, a system of dextral faults accommodates 15%–25% of the Pacific–North American plate motion. The northern Walker Lane in northwest Nevada and northeast California occupies the northern terminus of this system. This young evolving part of the plate boundary offers insight into how strike-slip fault systems develop and may reflect the birth of a transform fault. A belt of overlapping, left-stepping dextral faults dominates the northern Walker Lane. Offset segments of a W-trending Oligocene paleovalley suggest ∼20–30 km of cumulative dextral slip beginning ca. 9–3 Ma. The inferred long-term slip rate of ∼2–10 mm/yr is compatible with global positioning system observations of the current strain field. We interpret the left-stepping faults as macroscopic Riedel shears developing above a nascent lithospheric-scale transform fault.”
https://pubs.geoscienceworld.org/gsa/geology/article-abstract/33/6/505/103809/Kinematics-of-the-northern-Walker-Lane-An?redirectedFrom=fulltext
Ding!
Thanks! I was hoping you’d agree.
Over the years, I’ve truly enjoyed yer yammerin’s of our west coast tectonic settings and origins….especially your thoughts concerning the demise of our long-gone Farallon plate (not to ignore the many other subjects you’ve touched on).
Love this stuff! I majored in Geology at schools in the Basin and Range area, so most of my field trips were in these areas.
I’ve been wondering about the swarm in Idaho recently. It’s kind of close to some of the older hotspot centers.
Aftershocks are still continuing outside Boise/Challis after the M 6.5 back on March 31. …not atypical for Great Basin activity which often features prolonged aftershock sequences. The main shock was a complex strike-slip event on/in the well-known Sawtooth Fault zone, and not really related to any volcanic or magmatic activity.
Further south, the M6.0 outside Wells, Nv in 2008 (normal faulting due to crustal extension) produced aftershocks for many months.
I had a charter to Challis in the late 70’s just after a 6ish quake lots of aftershocks and the locals were not the least concerned.. Nature of living
in this part of the west..
Good post! Got me digging into Nevada geology immediately. An ever learnig expirience, this blog!
An image with the fault line in the area:
Source: https://twitter.com/rangefront/status/1261497007652265985/photo/1
When one flies over that area from the Oregon border south one can see the various mountain ranges, mesas,
and valleys. Faults everywhere. Read some years ago
that a geologist said the “Walker range country is cracked like a cheap mirror.” Which goes with what Craig H. posted earlier. Thanks for those images.
40 years ago today was the eruption of Mt. St. Helens.
The New York Times published a nice article to go with the anniversary:
https://www.nytimes.com/2020/05/18/science/mt-st-helens-eruption.html?action=click&module=News&pgtype=Homepage
The question to me is if there is any rifting. Reference: Sea of Cortez. It is the other end of hte same zipper if the hypothesis that San Andreas is too stuck and too bendy to serve as the continental edge.
Smithsonian has a really nice (and large and granular) map of volcanic hazards in California. The region in Nevada is next door to Mono Lake, Mono-Inyo craters and Long Valley. That whole area is geologically quite interesting. Also gorgeous to visit.
The aftershocks show quite nicely which fault ruptured. It indeed connects two faults. One of those two is the other end of the fault that suffered the Ridgecrest quake a year ago, 100 mile to the south. It seems possible the Nevada quake was a consequence of the Ridgecrest quake, and resolved stress caused by that event.
Check out the Twitter account of Jamie the Geologist: https://twitter.com/snowhorse420
He has some nice pictures of the ground deformation off the ‘Mina NV Earthquake’ as they call it. The ring fractures are a meter deep in some places…
The lack of a legend on this is unfortunate. What magnitude quakes are the different sizes of yellow circles? What are the smaller green dots off to one side of the Fish Lake Valley fault? What does the fault color (yellow, red, or black) signify? Unfortunately we’re left to guess. At least they didn’t forget the scale. 🙂
There is a legend on the big map I posted but it is not adjacent to the Mono Lake map. It is adjacent to the map of the state (the individual areas are zoom-ins).
Nice forensic volcanology article in Nature from April 2020 about a mysterious eruption in 1108-1110, previously attributed to Hekla.
https://www.nature.com/articles/s41598-020-63339-3#Abs1
Nice paper but the case overall is quite thin. Hekla’s eruption became famous and was definitely part of it. The tree rings which show cooling in Siberia but warming around the Atlantic suggest a role for the north atlantic oscillation – not all weather is volcanic. I think they need rather more data than they have to make a solid case.
Interesting whether or not tropical storm Arthur will make landfall in the US today. I expect not but it will be close. Although, moving along a submerged beach should perhaps not count as landfall anyway.
Arthur isn’t the one to worry about. It is Amphan in the Bay of Bengal.
https://www.wunderground.com/cat6/category-5-amphan-to-send-massive-storm-surge-into-eastern-india-and-bangladesh?cm_ven=hp-slot-1
Oh, this is serious. If things go badly the mortality could be immense.
This is not good. But the area is vastly better prepared than it was in the 1970’s. There are good warning systems in place and there are hurricane shelters. That will help. The article says that weakening will not change the tidal surge. That is not entirely true, since the part of the surge caused by the low pressure will get less when central pressure rises.
15,000 died in 1991, when the population was 100M, now its 160M.
Not good.
And the 1970 disaster was in the hundreds of thousands. River deltas, storm surges, and a growing population are a bad combination. I hope for the best. But they are better prepared than they were in the past.
https://www.bbc.co.uk/news/science-environment-52713131
https://www.bbc.co.uk/news/science-environment-52701935
I never tried boiling toothpaste, I’m not sure if I should…
My old geology lecturer used to describe the mantle as ‘flowing like toothpaste’
Did he mean or describe it, the mantel, as an “Non-Newtonian-Fluid” ?
https://en.wikipedia.org/wiki/Non-Newtonian_fluid?wprov=sfla1
4.2 27km WNW of Petrolia, CA 2020-05-19 15:58:31 (UTC) 23.1 km
Bit further away but also
4.4 260km W of Ferndale, California 2020-05-19 07:10:10 (UTC) 10.0 km
5.5 252km W of Ferndale, California 2020-05-18 07:02:34 (UTC) 10.0 km
4.7 255km W of Ferndale, California 2020-05-18 06:56:01 (UTC) 10.0 km
Mendocino triple junction. Interesting.
The Gordo microplate historically has the most activity around it’s periphery.
Update on Amphan:
https://www.wunderground.com/cat6/amphan-pushing-wind-rain-and-potentially-fierce-surge-into-eastern-india-and-bangladesh?cm_ven=hp-slot-1
It is nearing landfall.
That seems good news presented as bad news
How so?
Or … is the area experiencing a drought, such that it could really use the rain?
The report said that the hurricane was losing strength before landfall, and the speed of the trajectory is such that rain amounts would not be as high as could have been. I read that as good news, of course only relative to what was predicted earlier. A silver lining, if you wish.
The damage seems considerable, but casualties are so far limited.
Watch out for anticlockwise rain surges running up into Bangladesh as the warm moist air from the gulf is funneled inland up the Padma River.
New post is up! Bringing Laki to a close: how to light a fire.
https://www.volcanocafe.org/laki-the-making-of-a-fire/